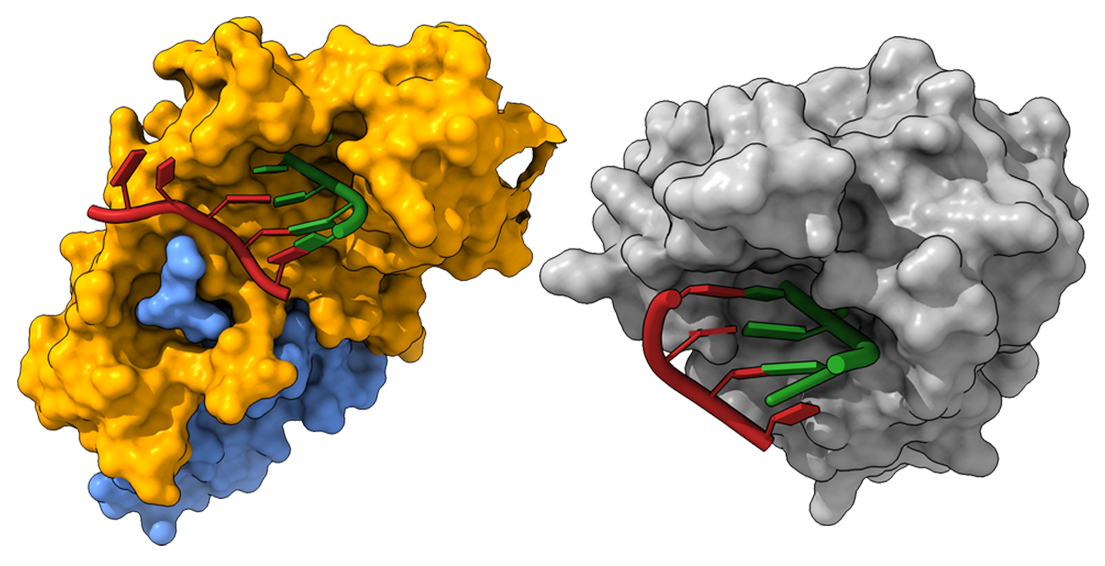
Project 4: Nuclease Inhibitors for Viruses of Pandemic Concern Abstract
SARS-CoV-2 (SARS2) and highly pathogenic arenaviruses, including Lassa virus (LASV), Junin virus, and Machupo virus, share structurally and functionally related enzymes that are capable of degrading an RNA strand. These enzymes, called exoribonuclease (ExoN), play essential roles in proofreading viral RNA and suppressing antiviral responses. The proofreading capabilities of SARS2 have shown that the virus is effective at stably self-correcting errors in its genome.
Project 4 is pursuing chemical inhibition to block viral replication by lethal mutagenesis (loading up the virus with deleterious mutations) and slowing viral infection by reactivating immune responses.
Our team has contributed extensively to understanding these viral ExoN enzymes, including shedding light on their atomic structures and catalytic mechanisms. We have also developed robust fluorescence-based tests to analyze ExoN activities, including a novel method that enables a gain-of-function readout in ultra-high-throughput screening (uHTS) of small molecule inhibitors. Furthermore, we have used DNA-encoded chemistry technology and computational screening to target SARS2 ExoN and LASV ExoN.
Building on these prior and preliminary studies, we will identify first-in-class viral ExoN inhibitors. We will then leverage medicinal chemistry and structural biology to enhance compounds' potency, selectivity, and biochemical and physiologic properties for antiviral studies in cell and animal models. These studies will deliver antiviral drug candidates with a distinct mechanism of action to complement those developed against established antiviral targets, helping to address the critical need for novel antiviral drugs against both SARS2 and the highly pathogenic arenaviruses that cause fatal hemorrhagic fever infections.